 If you’re like me, sometimes you want to silence all Dexcom alarms.
If you’re like me, sometimes you want to silence all Dexcom alarms.
Maybe you’re at church, a wedding, or a funeral. Possibly a job interview or an important meeting. Maybe you just want to sleep. Maybe it is the first 24 hours of a sensor with erratic readings and nosediving lows.
All Dexcom alerts except for the 55-Low alarm can be turned off on both your phone and Tandem pump. But you can’t turn off the 55-Low critical alarm or even pause it for a certain period of time. Some people think that is a great safety feature and have no problems with the limitation. Me, I struggle with it. Although Dexcom is a great tool and I appreciate that it regularly gives me notifications of highs and lows, I don’t believe that it saves my life every day. I am confident that if I can silence all Dexcom alarms at certain times, I am going to be okay. Sometimes my diabetes mental health requires that. I don’t want my medical device to siren at my niece’s wedding or during an important business meeting. (Actually I am retired and don’t have important business meetings. But you know what I mean.)
In August I wrote about my frustrations with Day 1 Dexcom sensors. Many or most of my sensors start at a number much lower than my actual meter-confirmed BG. While it is easy to turn off Control IQ on my Tandem pump until the sensor is reliable, the stress of repeating false 55-Low alarms on both my phone and pump is sometimes unbearable. I want to STFU those alarms! Not forever. Just for a couple of hours until the sensor stabilizes. Here is a reminder photo from that August blogpost.

When I am losing the battle with Dexcom false low alarms, I can turn off my phone for a while or overnight if I am not expecting important phone calls. But the pump is different. I can’t turn it off or bury it in my sock drawer because I need insulin delivery. I can’t pause or completely silence the sensor on the pump without stopping the sensor session. I am stuck.
It recently dawned on me that there is a way to silence the 55-Low alarm on my pump until the sensor is stable and reading accurately. I recently purchased a small Faraday bag which magically prevents Bluetooth and other electronic signals from communicating with whatever device I put in the bag. The Faraday pouch allows me to keep my pump running without turning off the active sensor session. But it stops the transmitter from sending false low numbers to the pump and prevents the incessant low-55 alarms. (FYI you must turn off the Out-of-Range alarm for the period that the pump is in the Faraday bag or you’ll get that alarm. Options/My CGM/CGM Alerts/Out of Range.) If I am concerned that I might have a real low, I use my meter. For something short like a funeral, a real low is an unlikely occurrence as long as my BG starts at a good number. For a sensor startup, I’ll probably use my meter several times whether or not my pump is in a Faraday bag.
If you don’t know about Faraday bags, you can do an internet search and learn about how they work. People use this technology to protect key fobs, credit cards, phones, and other electronic devices from remote hacking. Scientists have lots of exotic uses for Faraday cages that block all electromagnetic interference. Me, I just want it to temporarily isolate my pump from inaccurate sensor readings.
If you’re someone who doesn’t struggle with Dexcom inaccurate numbers and isn’t bothered by alarms, a Faraday pouch is probably not for you. But if you are like me and sometimes just want to be protected from alarms that are inaccurate and/or intrusive, you might want to give a Faraday bag a try. There are lots of choices online and I purchased this one for about $20 through Amazon. I wasn’t convinced that the Extra-Small size would work (although I think it would), so I bought Small. It is big enough for my phone and definitely roomy for the pump.
When you remove your pump from the Faraday bag, initially it will show the Out of Range symbol. It takes 5 minutes or less for the pump to reconnect to the Dexcom transmitter.

Some people will appreciate the ideas in this blogpost. Others, especially parents of young children and people not diagnosed in the dark ages, will be askance at the idea of blocking Dexcom signals.
But me, sometimes I need my diabetes tech to leave me alone. I am not a total rebel and mostly I do a good job of managing my blood sugar and staying safe. But sometimes I am just an imperfect human trying to deal with imperfect D-tech and I need a way to protect my mental health. The Faraday bag is a new tool in my arsenal.
*********
Note: The Faraday bag is not a solution for the siren of Tandem occlusion alarms. Fortunately with my recent pumps, I am rarely getting those alarms anymore. But I have memories of shrieking (false) alarms in yoga class and at the movies. So far none at a wedding or funeral….

 I was given the opportunity to read and review Stacey’s second book, Still The World’s Worst Diabetes Mom. In general I turn down most opportunities of free stuff because although I usually write favorable reviews, I rarely (or never) continue to use the reviewed product. It seems a bit fraudulent to advertise things to my readers that I don’t use. But books are different. It is a rare book about diabetes that I don’t learn from or at least get inspiration from. Plus I want to support the authors of the Diabetes Online Community (DOC).
I was given the opportunity to read and review Stacey’s second book, Still The World’s Worst Diabetes Mom. In general I turn down most opportunities of free stuff because although I usually write favorable reviews, I rarely (or never) continue to use the reviewed product. It seems a bit fraudulent to advertise things to my readers that I don’t use. But books are different. It is a rare book about diabetes that I don’t learn from or at least get inspiration from. Plus I want to support the authors of the Diabetes Online Community (DOC).

 The title of this blogpost might indicate that I think Covid has gone away and is no longer a risk.
The title of this blogpost might indicate that I think Covid has gone away and is no longer a risk. Covid seriously with masks and vaccines. Even still, three of the four parents have had the virus in the last 6 months as have six of the seven grandchildren.
Covid seriously with masks and vaccines. Even still, three of the four parents have had the virus in the last 6 months as have six of the seven grandchildren. I started Duolingo Spanish in 2020 and am now at Day 752 of daily Spanish lessons. I have learned a lot but am miles away from being a fluent Spanish speaker. I am currently on Unit 50 with Unit 211 as the end goal. I can read many things in Spanish and understand random words from Spanish speakers. I occasionally stumble through a sentence or two with my grandchildren’s nanny from South America.
I started Duolingo Spanish in 2020 and am now at Day 752 of daily Spanish lessons. I have learned a lot but am miles away from being a fluent Spanish speaker. I am currently on Unit 50 with Unit 211 as the end goal. I can read many things in Spanish and understand random words from Spanish speakers. I occasionally stumble through a sentence or two with my grandchildren’s nanny from South America.
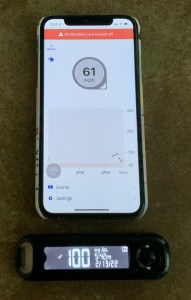
 Every ten days my diabetes mental health is in jeopardy.
Every ten days my diabetes mental health is in jeopardy. Or maybe I am lucky and get a number. I take a BG test with my meter. 94
Or maybe I am lucky and get a number. I take a BG test with my meter. 94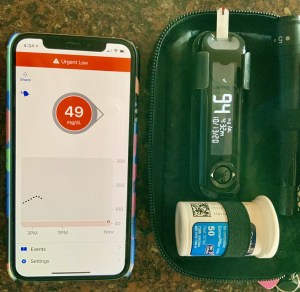

 I probably write about Control IQ more often than I should. Some people with Tandem pumps love it and others hate it. I never love it but I often appreciate the benefits of a computer algorithm helping me out with my diabetes. Unfortunately I sometimes think that Control IQ
I probably write about Control IQ more often than I should. Some people with Tandem pumps love it and others hate it. I never love it but I often appreciate the benefits of a computer algorithm helping me out with my diabetes. Unfortunately I sometimes think that Control IQ
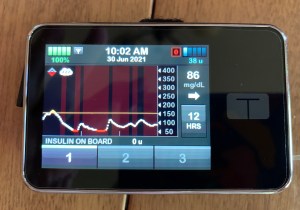

 diabetes strive for?
diabetes strive for?